Introduction
Newborns feel pain (1, 2). Repeated painful stimuli lower their pain threshold (3, 4) by overstimulation of NMDA receptors, which may lead to excitotoxic brain damage (5). Until a few years ago, it was claimed that the word pain was inappropriate for newborns, as pain is a subjective experience that newborns, because of their age, cannot have (6). Until the 1980s, analgesics were rarely administered to newborns even in the case of surgery (7). Now we know that anesthesia reduces brain damage due to hypoxemia, hypertension, tachycardia, variations in heart rate, and increased intracranial pressure (8, 9), all of which are particularly dangerous because of immature cerebral vasoregulation in the premature (10).
The number of painful stimuli needs to be kept to a minimum, and every effort should be made to render them less painful. Guidelines for neonatal analgesia have been suggested (11–15), especially for the most routine type of pain, blood sampling, which is usually performed by heel prick. To avoid the drawbacks of general and local analgesics (16–21), types of nonpharmacologic analgesia have been proposed, including nonnutritional sucking and instillation of glucose or other sweet liquids on the newborn’s tongue (22). The analgesic effect of glucose is thought to stimulate an increase in plasma concentrations of β-endorphin (23–27) by a preabsorptive mechanism (28).
Sensory stimuli combined with oral glucose during heel prick greatly reduced manifestations of pain in preterm babies (29). This combination was even more effective in premature babies than oral glucose. It was called sensorial saturation (SS) because it works through competition between nonpainful and painful stimuli, not to indicate that nonpainful stimuli cause saturation of sensorial pathways. In the present study, we investigate whether SS promotes analgesia in term babies and whether it is more effective than that obtained with sugar alone.
Material and methods
The retrospective study was performed in the San Filippo Neri Hospital, Rome (Italy). In our hospital babies routinely underwent two types of non-pharmacological analgesia when undergoing heelprick for blood sampling: oral 12% sucrose or sensorial saturation. Sensorial saturation consists of a multisensory stimulation composed of sweet taste (12% oral sucrose), touch (massaging the baby’s face) and talk (voice is used to distract the baby). Sucrose is instilled on the tip of the tongue with a sterile syringe, introduced in the infant’s mouth and moved to stimulate sucking. The amount of liquid, administered drop by drop, is that necessary to maintain sucking until after the heel-prick, i.e. 1 ml 2 minutes before heelprick. Sensorial saturation is administered as follows. After administering oral sucrose, the nurse began talking to the baby and massaging its face to distract him/her; two minutes after the beginning of these manoeuvres, heel prick was performed.
We usually give a pain score to all babies during these procedures using the NIPS scale, a validated pain scale scoring from 0 (no pain) to 7 (maximum pain).
We retrospectively included in this study all babies with the following features: (1) postconceptional age >35 weeks; (2) intraventricular hemorrhage (IVH) Mean scores in the two study groups were statistically compared using the Student’s t test.
Results
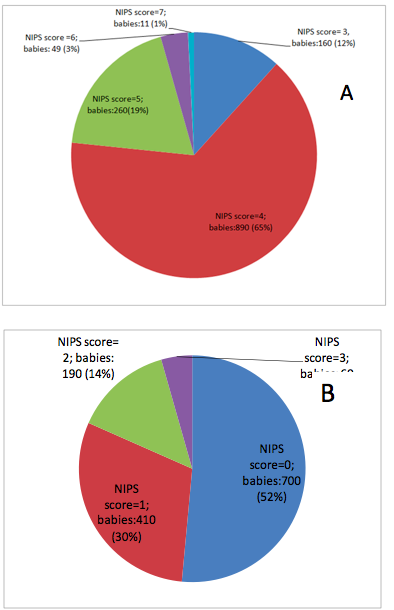
Babies who fulfilled the inclusion criteria were divided into two groups of 1360 babies each: one receiving oral 12% sucrose and one receiving Sensorial Saturation. The main pain scores of the two groups were respectively 4.16 (SD=0.64) and 0.71 (SD=0.84), with a statistically significant difference (p<0.001). Figure 1 reports the rate of presence in both groups (oral sucrose or sensorial saturation), of babies with distinct grades of NIPS scores.
Discussion
This is the widest study ever conducted in the field of nonpharmacological analgesia in neonates, and the first that compares sensorial saturation with oral sucrose. The evidence that sensorial saturation is more effective than oral sucrose results from the statistical comparation and from the rate of high pain scores in the two study groups. These data confirm previous studies on the effectiveness of sensorial saturation. With SS, we found an almost complete absence of pain reaction during heel prick; the babies rarely cried, and if they did, only for a few seconds. Other researchers (30-34) succeeded in reducing crying of premature babies with oral sucrose, but the infants nevertheless cried for many seconds and in some cases for more than 1 min, much more than with our technique, which reduced the mean crying time in the 30 s after heel prick to 2.8 s. Haouari et al. (34) were on the right track when they wrote, “We don’t know whether simply cuddling an infant after heel prick is as effective in reducing crying as 50% sucrose.” In actual fact, concentrated sugar solution is much more effective than cuddling, but the latter or at least its sensory component, i.e. massage, voice, rocking, eye fixing, adds further analgesia to the analgesic effect of glucose and provides a human factor in the form of company to a baby facing pain. SS is a technique that can be used for all newborns undergoing blood samples or other minor painful procedures. This study included a high number of babies, and it is one of the most numerous studies in this field, with a consequent high strength. A major limitation is its retrospectiveness and the fact that it could not be blinded, since the nurse who scored pain was present at the procedure and could not ignore the type of analgesia the infant received.
More studies are necessary to assess the best analgesic treatment for babies during procedural pain; this study is an important benchmark.
References
1. Anand KJ, Hickey PR. Pain and its effects in the human neonate and fetus. N Engl J Med 1987; 317: 1321-1326.
2. Fitzgerald M, Millard C, MacIntosh N. Hyperalgesia in premature infants. Lancet 1988; 1: 29.
3. McIntosh N. Pain in the newborn, a possible new starting point. Eur J Pediatr 1997; 156: 173–177.
4. Menon G, Anand KJ, McIntos. Practical approach to analgesia and sedation in the neonatal intensive care unit. Semin Perinatol 1998; 22: 417-424.
5. Anand KJ, Scalzo FM 2000 Can adverse neonatal experiences alter brain development and subsequent behavior? Biol Neonate 2000; 77: 69-82.
6. Richard T 1985 Can a fetus feel pain? BMJ 1985; 291:1220-1221.
7. Beyer JE, DeGood DE, Ashley LC, Russell GA 1983 Patterns of postoperative analgesic use with adults and children following cardiac surgery. Pain 1983; 17:71-81.
8. Anand KJ. Clinical importance of pain and stress in preterm neonates. Biol Neonate 1988; 73: 1-9.
9. Stevens BJ, Johnston CC. Physiological responses of premature infants to a painful stimulus. Nurs Res1994; 43: 226-231.
10. Tsuji M, Saul P, du Plessis A, Eichenwald E, Sobh J, Crocker R, Volpe JJ. Cerebral intravascular oxygenation correlates with mean arterial pressure in critically ill premature infants. Pediatrics 2000; 106: 625-632.
11. Canadian Pediatric Society. Fetus and Newborn Committee 2000 Prevention and management of pain and stress in the neonate. Pediatrics 2000; 104: 454-458.
12. Spaeth JP, O’Hara IB, Kurth CD. Anesthesia for the micropremie. Semin Perinatol 1998; 22: 390-401.
13.Stevens B, Gibbins S, Franck LS. Treatment of pain in the neonatal intensive care unit. Pediatr Clin North Am 2000; 47: 633-640.
14. Carbajal R, Simon N. Sédation et analgésie chez l’enfant. Arch Pediatr 1995; 2: 1089-1096.s
15. Anand KJ. Consensus statement for the prevention and management of pain in the newborn. Arch Pediatr Adolesc Med 2001; 155: 173-180.
16. Suresh S, Anand KJ. Opioid tolerance in neonates: mechanisms, diagnosis, assessment, and management. Semin Perinatol 1998; 22: 425-433.
17. Jacqz-Aigrain E, Burtin P. Clinical pharmacokinetics of sedatives in neonates. Clin Pharmacokinet 1996; 31: 423-443.
18. Levene M. Pain relief and sedation during neonatal intensive care. Eur J Pediatr 1995; 154: S22-S23.
19. Law RMT, Halpern S, Martins RF, Reich H, Innanen V, Ohlsson A. Measurement of methemoglobin after EMLA analgesia for newborn circumcision. Biol Neonate 1996; 70: 213-217.
20. Gourrier E, Karoubi P, El Hanache A, Merbouche S, Mouchnino G, Dhabhi S, Leraillez J. Utilisation de la crème EMLA chez le nouveau-né à terme et prématuré. Etude d’efficacité et de tollerance. Arch Pediatr 1995; 2: 1041-1046.
21. Lemmen RJ, Semmekrot BA 1996 Muscle rigidity causing life-threatening hypercapnia following fentanyl administration in a premature infant. Eur J Pediatr 155:1067.
22. Blass EM. Milk-induced hypoalgesia in human newborns. Pediatrics 1997; 99: 825-829.
23. Balon-Perin S, Kolanowski J, Berbinschi A, Franchimont P, Ketelslegers JM. The effects of glucose ingestion and fasting on plasma immunoreactive beta-endorphin, adrenocorticotropic hormone, and cortisol in obese subjects. J Endocrinol Invest 1991; 14: 919-925.
24. Tropeano G, Lucisano A, Liberale I, Barini A, Vuolo IP, Martino G, Menini E, Dell’Acqua S. Insulin, C-peptide, androgens, and beta-endorphin response to oral glucose in patients with polycystic ovary syndrome. J Clin Endocrinol Metab 1994; 78: 305-309.
25. Blass EM, Fitzgerald E. Milk-induced analgesia and comforting in 10-day-old rats: opioid mediation. Pharmacol Biochem Behav 1988; 29: 9-13.
26. Shide DJ, Blass EM. Opioidlike effects in intraoral infusions of corn oil and polycose on stress reactions in 10-day-old rats. Behav Neurosci 1989; 103: 1168-1175.
27. Blass E, Fitgerald E, Kehoe P. Interactions between sucrose pain and isolation distress. Pharmacol Biochem Behav 1987; 26: 483-489.
28. Ramenghi LA, Evans DJ, Levene MI 1999 “Sucrose analgesia”: absorptive mechanism or taste perception? Arch Dis Child Fetal Neonatal Ed 1999; 80: F146-F147.
29. Bellieni CV, Buonocore G, Nenci A, Franci N, Cordelli DM, Bagnoli F. Sensorial saturation: an effective analgesic tool for heel-prick in preterm infants. A prospective randomized trial. Biol Neonate 2001; 80: 15-18.
30.Overgaard C, Knudsen. Pain relieving effect of sucrose in newborns during heel prick. Biol Neonate 1999; 75: 279-284.
31. Bucher HU, Baumgartner R, Bucher N, Seiler M, Fauchère JC. Artificial sweetener reduces nociceptive reaction in term newborn infants. Early Hum Dev 2000; 59: 51-60.
32. Ramenghi LA, Griffith GC, Wood CM, Levene MI. Effect of non-sucrose sweet tasting solution on neonatal heel prick responses. Arch Dis Child 1996; 74: F129-F131.
33. Skogsdal Y, Eriksson M, Schollin J. Analgesia in newborns given oral glucose. Acta Paediatr 1997; 86: 217-220.
34. Haouari N, Wood C, Griffiths G, Levene M. The analgesic effect of sucrose in full term infants: a randomised controlled trial. BMJ 1995; 310: 1498-1500.
